Endoscopic Submucosal Dissection for Treatment of Localized Gastric Mucosa-associated Lymphoid Tissue Lymphoma: A Case Series
Article information
Abstract
Background/Aims
The treatment for gastric mucosa-associated lymphoid tissue lymphoma (MALToma) generally involves eradication of Helicobacter pylori. However, MALToma lesions may recur even without H. pylori re-infection. Furthermore, the remission rate of H. pylori-negative MALToma after eradication is low. Therefore, herein, we report on endoscopic submucosal dissection (ESD) as a treatment strategy for gastric MALToma.
Methods
We retrospectively reviewed the data of all patients of gastric MALToma who underwent endoscopic resection at our institution between January 2000 and December 2021. Clinical remission was defined as complete histological remission or probable minimal residual disease according to the GELA grading system for post-treatment evaluation of gastric MALToma.
Results
Six patients with gastric MALToma underwent ESD. Two patients were diagnosed with gastric MALToma, which improved after eradication treatment and relapsed approximately 36 and 41 months later, respectively. These patients had singular lesions localized to the mucosa and did not experience H. pylori re-infection. The lesions were successfully removed via ESD. The remaining four patients had H. pylori-negative gastric MALToma. These patients also had single, localized lesions that were removed via ESD. All the patients remained in clinical remission until the final follow-up.
Conclusions
ESD is a safe and effective intervention for H. pylori-negative gastric MALToma when the lesion is single and confined to the mucosal layer.
INTRODUCTION
A gastric mucosa-associated lymphoid tissue lymphoma (MALToma) is an extranodal marginal zone lymphoma and the most common primary gastric lymphoma [1]. It is also known that Helicobacter pylori infection plays an important role in the development of this cancer [2,3]. H. pylori eradication is typically therefore the first treatment method for gastric MALToma regardless of stage [4,5]. A systemic review in 2009 reported a complete histological remission (CR) rate of 973 out of 1,250 patients that underwent eradication therapy for H. pylori-positive gastric MALToma [6]. By contrast, another systematic review indicated a CR rate of only 15.5% in patients with H. pylori-negative gastric MALT lymphoma with eradication therapy [7]. Recurred gastric MALToma cases after CR can be treated with eradication therapy alone, or in combination with radiation therapy, if they are H. pylori-positive [8]. As H. pylori eradication therapy will not be effective in recurred cases without H. pylori infection, second-line treatments such as radiotherapy or chemotherapy may be helpful with achieving a CR status in these cases [8,9].
Some recent case reports have indicated that CR can be achieved in localized gastric MALTomas treated with endoscopic submucosal dissection (ESD) [10,11]. Herein, we describe our case series of localized gastric MALTomas that were successfully treated with ESD.
MATERIALS AND METHODS
1. Patients
We retrospectively investigated patients who underwent either gastric endoscopic mucosal resection (EMR) or ESD at Asan Medical Center (Seoul, Korea) between January 2000 and December 2021 (Fig. 1). Twenty-three patients were diagnosed with gastric MALToma via a pathologic assessment after the endoscopic resection. We excluded patients who underwent endoscopic resection for diagnostic purposes. Among them, 6 patients who underwent ESD as a curative intervention for gastric MALToma were included in this present study cohort. The patients’ symptoms, medical histories, H. pylori infection status, pathologic results, and endoscopic findings were reviewed from the electronic medical records. This study was approved by the Institutional Review Board of Asan Medical Center (approval number: 2022-0082). Informed consent was waived by the IRB.

Flowchart of the study with gastric MALToma who underwent endoscopic resection. EMR, endoscopic mucosal resection; ESD, endoscopic submucosal dissection; MALToma, mucosa-associated lymphoid tissue lymphoma. aOther cancers included hyperplastic polyps (n=2), early gastric cancer (n=5), tubular adenoma (n=2), and gastrointestinal stromal tumor (n=1).
2. Endoscopic procedure
All six study patients were admitted to the hospital one day before the procedure. All ESD procedures were performed by two experienced operators (C.K.D. and J.H.Y.) using a single-channel scope with insulation-tipped (IT), IT-2, hook, and dual knives. Patients were sedated using intravenous midazolam (0.05 mg/kg) with pethidine (25 or 50 mg) immediately before the procedure. Marker dots encircling the lesion at an adequate distance were denoted and the lesions were elevated via a submucosal injection of saline solution combined with epinephrine (0.01 mg/mL) and indigo carmine. The circumference of the raised lesions was then cut off and a submucosal dissection was performed. If post-procedural active bleeding or an exposed vessel were detected, preemptive endoscopic hemostasis was performed on the oozing or exposed vessels. The specimens were retrieved using an endoscopic net.
3. Definitions
The pathologic diagnosis of a gastric MALToma in each of the study subjects was based on the resected specimen obtained after ESD. En bloc resection was defined as removal of the ESD specimen in a single piece without separation. Complete resection was defined as resection of the tumor without deep or lateral margin involvement of the gastric MALToma. Clinical remission was defined as either CR or probable minimal residual disease (pMRD) in accordance with the Group d’Etude des Lympohmes de I’Adult (GELA) grading system for the post-treatment evaluation of gastric MALTomas [12,13].
RESULTS
The six included study patients with gastric MALToma were treated with ESD as a curative intervention at Asan Medical Center between 2008 and 2020 (Fig. 2). The rationale for conducting the ESD procedure in these cases included a recurrence of gastric MALToma without H. pylori re-infection in two cases (Cases 1 and 2), and a H. pylori-negative MALT lymphoma in the other four patients (Cases 3~6). The baseline characteristics of the abovementioned patients are summarized in Table 1 and clinical outcomes of ESD, and histopathologic findings are presented in Table 2. Two representative cases are detailed below.
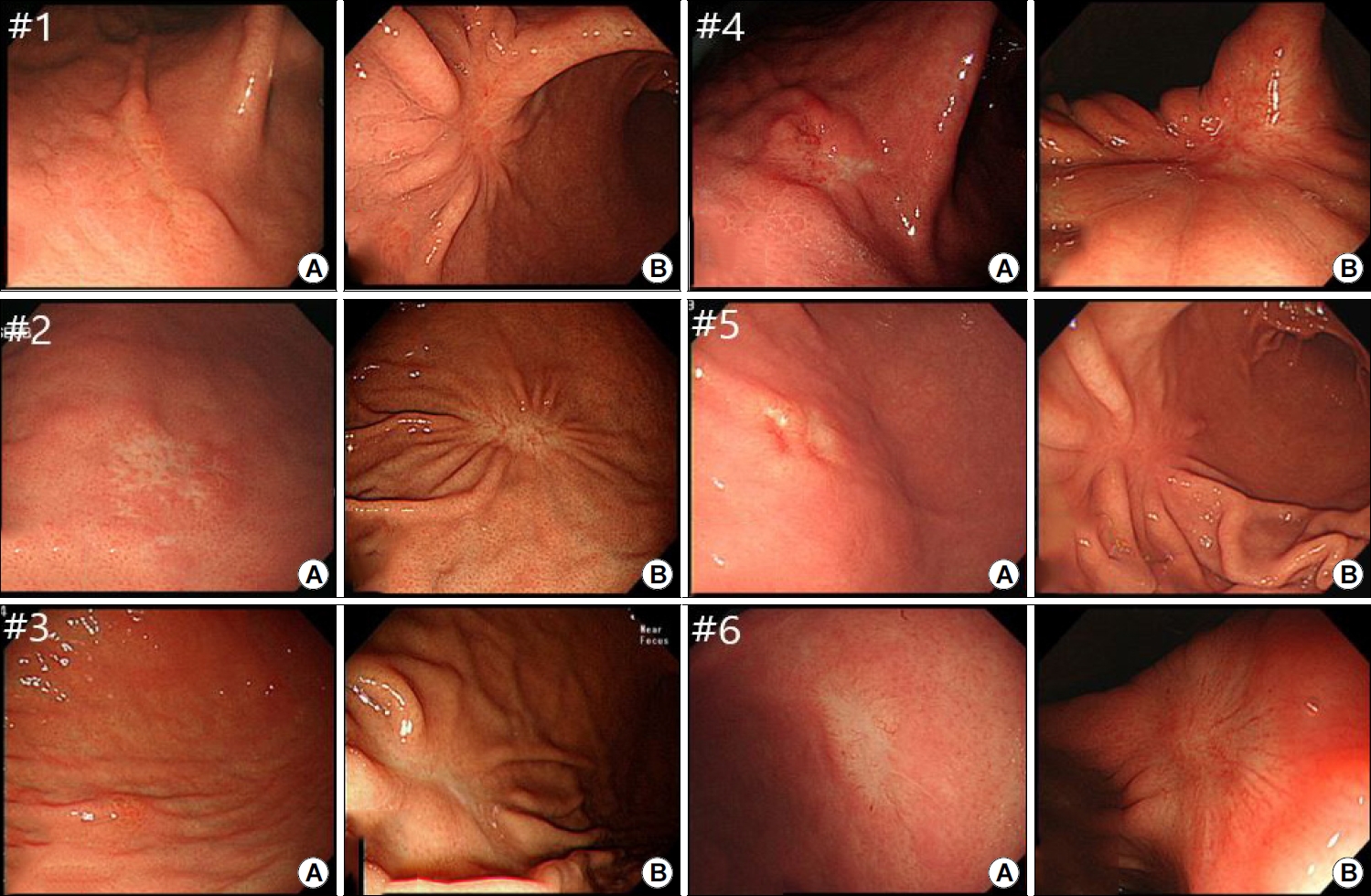
Endoscopic images of the six study patients. (A) Representative endoscopic images of gastric MALTomas prior to ESD. (B) Representative endoscopic images of ESD-induced ulcer scarring without evidence of gastric MALToma recurrence.ESD, endoscopic submucosal dissection; MALToma, mucosa-associated lymphoid tissue lymphoma.

Baseline Characteristics of the Six Study Patients with Gastric Mucosa-associated Lymphoid Tissue Lymphoma Treated via Endoscopic Submucosal Dissection

Clinical Outcomes after Endoscopic Submucosal Dissection for Localized Gastric MALToma in the Six Study Patients
1. Case 1: recurred gastric MALToma without H. pylori re-infection
A 60-year-old man with a recurred gastric MALToma presented at our hospital. He underwent an EGD as a medical check-up in December 2010 and was diagnosed at that time with localized gastric MALToma at the great-curvature side of lower body with H. pylori infection. After receiving a first line eradication treatment, the patient had been maintained in CR but the gastric MALToma recurred at the same site about 3 years later without any symptoms on a follow-up EGD. Accordingly, the patient was referred to our hospital and was admitted for further evaluation. A urea breath test, rapid urease test, and H. pylori IgG antibody assay all showed negative results. Imaging assessments of the patient including computed tomography (CT) and positron emission tomography (PET) scans indicated no other organ involvement. A bone marrow biopsy also showed no disease involvement. Accordingly, we decided to perform an ESD procedure because it was thought that this patient’s lesion was localized and confined to the mucosa. The lesion was subsequently resected en bloc and the patient was discharged without any complications. Analysis of the resected specimen revealed complete resection of the gastric MALToma. Annual surveillance endoscopies have since found only ESD-induced ulcer scarring without any further recurrences over 85 months.
2. Case 3: gastric MALToma with out H. pylori infection
A 61-year-old woman without any symptoms was diagnosed with gastric MALToma at the greater curvature of the lower body during a medical check-up in December 2014. She had a history of receiving a first eradication treatment for H. pylori infection in 2008. The patient was admitted to our hospital for further evaluation by urea breath test, rapid urease test, and H. pylori IgG antibody testing but all were negative. Imaging analysis including CT and PET scans and a bone marrow exam revealed no evidence of organ involvement of the lymphoma. The patient received another H. pylori eradication treatment (sequential therapy) despite a negative test result for this bacterium and her progress was monitored. She did not improve however, even after 42 months. We thus decided to perform ESD as a curative purpose. The lesion was dissected clearly using this technique and the patient was discharged without any complications. The resected specimen revealed complete resection of the gastric MALToma. Subsequent annual follow-up endoscopies have indicated that a CR status had been maintained at 44 months post-procedure.
3. Pathologic findings
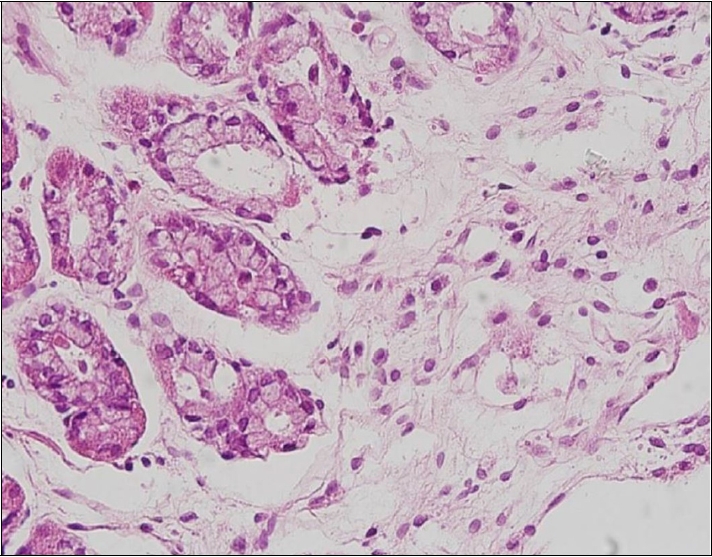
Histologic examinations including immunohistochemistry were conducted for all specimens. Five specimens (all except Case 4) underwent en-bloc resection and showed dense, mass-lymphocytic infiltration, which was centered in the deep mucosa and expanded to the mucosal surface and submucosa with a clear resection margin evident under a low power view (Fig. 3A). In a higher power view, small monomorphic lymphocytes were visible and had caused damage to the gastric glands (Fig. 3B, arrow). These infiltrating lymphocytes were diffusely stained for CD20, supporting the diagnosis of gastric MALToma (Fig. 3C). Case 4 also showed similar histologic findings, but the infiltration was deeper with this patient than for the other cases and the deep resection margin had MALToma involvement (Fig. 4). On follow-up biopsies, in accordance with GELA staging, five patients (all except Case 4) had a CR status (Fig. 5) and Case 4 had pMRD (Table 2).
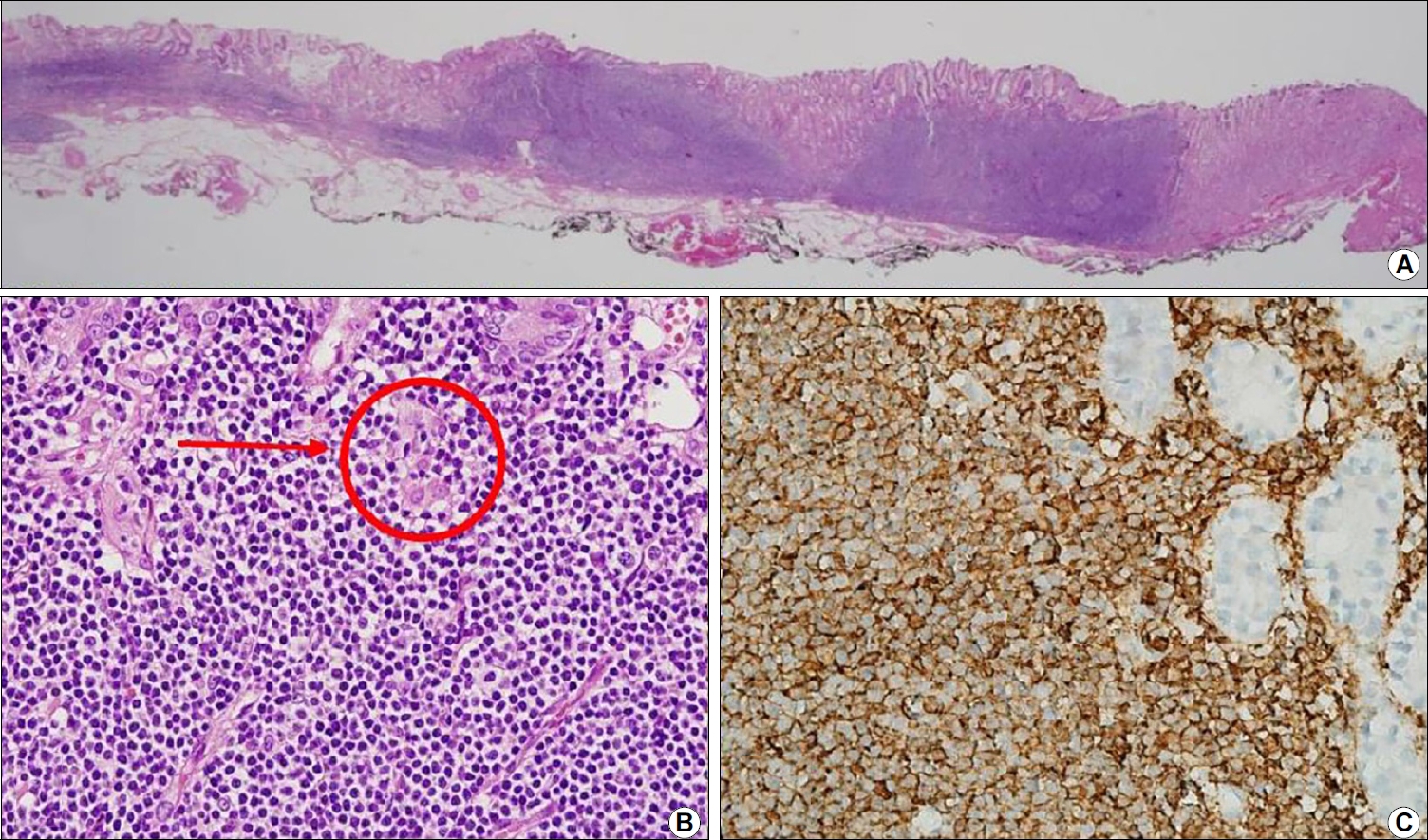
Representative histopathological images of the resected and follow-up biopsy specimens of the study patients. (A) Low-power view of an endoscopic submucosal dissection specimen showing dense lymphocytic infiltration centered in the deep mucosa and expanding to the mucosal surface and submucosa (H&E, ×12.5). (B) High-power view of the tissue is shown in (A) showing small monomorphic lymphocytes damaging the gastric glands (arrow) (H&E, ×400). (C) Infiltrating lymphocytes were diffusely stained for CD20, supporting the diagnosis of MALToma (immunohistochemistry, ×400).
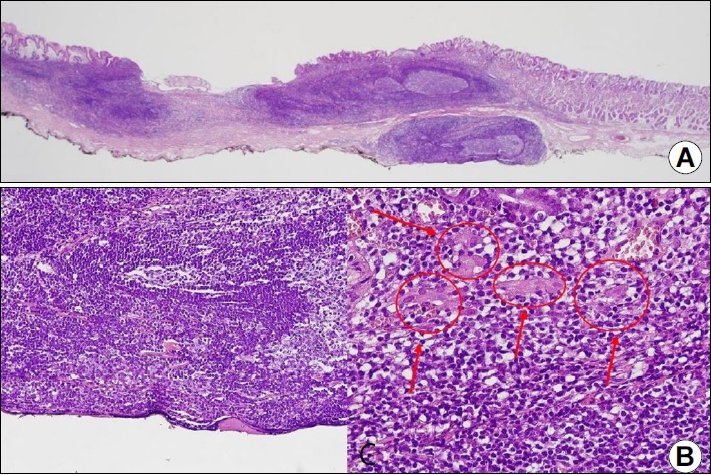
Representative histopathological images of the resected specimen in Case 4. (A) Low-power view of the tissue obtained after endoscopic submucosal dissection showing a dense, mass-like lymphoproliferative lesion. The lesion contained some follicles and the marginal zones were expanded, with deep submucosal invasion (H&E ×12.5). (B) The deep resection margin for this lesion (H&E, ×200). (C) Infiltrating lymphocytes were small and monomorphic and had damaged the gastric glands (arrows) (H&E, ×400).
DISCUSSION
Gastric MALToma can present in various forms at any location but usually arise in the body of the stomach [14]. While these cancers can develop as localized single lesions within the tissue of origin with various morphologies, previous studies have reported multiple gastric involvement in 30.0~47.7% of cases [14-16], so that detailed observations during EGD, and multiple biopsies, are needed to properly determine the region of involvement. Detecting the presence of H. pylori infection is also very important in patients with gastric MALToma because the absence of this bacterium is known as a predictive factor for resistance to eradication therapy [7,17]. Hence, a variety of diagnostic methods are recommended to definitively exclude any false-negative results from H. pylori testing [18]. Accordingly, we confirmed the H. pylori infection status by at least two methods among our own patients, including a urea breath test, rapid urease test, and H. pylori IgG antibody test.
Among the six patients we analyzed in our present study, two cases (Cases 1 and 2) had a recurred gastric MALToma without H. pylori re-infection. A previous study reported a 3.1% rate of relapse after CR in patients with gastric MALToma (10/323 patients), of which 6 were treated with chemotherapy and radiation therapy [9]. The national cancer center network guidelines recommend radiation therapy for cases of recurrent gastric MALToma without H. pylori infection [8]. Four patients in our present study series (Cases 3~6) were gastric MALToma cases without H. pylori infection for which the treatment recommendations are radiation therapy or rituximab as the first-line intervention for stage I or II1 lesions [8]. Hence, we could consider radiation therapy in our six study subjects. However, since the lesions in each of these patients was single and localized, we utilized the ESD technique to successfully achieve a CR status.
The incidence of bone marrow involvement of a gastric MALToma is relatively low, reported at approximately 0.5~6.7% [14,19,20]. It is also not certain how such an involvement would affect the disease prognosis [20,21]. Won and colleagues previously reported that prognosis of gastric MALToma patients with subtle CD20 positivity in the bone marrow exam was not significantly affected [22]. As our current case 2 also showed bone marrow involvement with only subtle CD20 positivity and CD3 negativity on immunohistochemistry, we considered that the patient’s prognosis would not be affected and performed a successful ESD leading to a maintained CR status in this patient at the time of writing.
Involved-field radiation therapy could be the preferred option for a localized gastric MALToma in patients who do not achieve remission after eradication therapy [23]. Excellent disease control using moderate-dose radiation therapy at the stomach and perigastric nodes over 3~4 weeks has been described in multinational studies [24,25]. Anorexia, nausea, and vomiting are common acute toxicities from this intervention, but are mild in most instances. Although long-term toxicity is infrequent following radiation therapy, an increased incidence of secondary malignancy after a radiation protocol has been reported [25]. On the other hand, ESD has an acceptably low risk of acute complications such as bleeding and perforation. The advantage of ESD over radiation therapy includes shorter treatment periods and no long-term toxicity. We found in our current series that the patients underwent an ESD procedure safely and efficiently with no significant post-operative complications.
Pathologic evaluations of the resected specimens in our current cohort revealed a submucosal involvement of the gastric MALToma in all six patients. On EUS, four patients (Cases 2, 3, 5, and 6) were diagnosed with tumors localized at the mucosa, suggesting that the accuracy of this method is lower for gastric MALT lymphoma than for gastric adenocarcinoma. The explanation for this is that unlike adenocarcinomas, MALTomas do not involve a desmoplastic reaction or vascular proliferation in the underlying tissue, but instead infiltrate ‘silently’. It may be difficult therefore to detect a submucosal involvement of a MALToma by EUS unless the tumor had a sufficiently deep invasion or formed a distinct mass-like lesion. After ESD, although five of our current study patients underwent resection with a clear margin, the remaining patient (Case 4), who showed submucosal involvement on EUS, had a deep margin involvement of the gastric MALToma. In this patient, we could have considered radiation therapy to remove the remaining lesions in the submucosal layer. However, 1) since final pathology showed a focally positive deep margin, the cauterizing effect of ESD could remove the residual tumor, 2) endoscopy and EUS showed a very low probability of proper muscle invasion, and 3) the patient was asymptomatic. Therefore, we discussed further radiotherapy and surveillance with the patient in detail and decided on surveillance.
There were some notable limitations to this study. First, in the gastric MALToma cases which was initially diagnosed and treated at another hospital, no previous specimen was available for a tissue review. Second, the follow-up period after the ESD was not sufficient in some cases.
In conclusion, ESD is a safe and effective method for treating patients with a gastric MALToma when the lesion is single and confined to the mucosal layer, and the patient is H. pylori-negative.
Notes
AVAILABILITY OF DATA AND MATERIAL
The datasets generated or analyzed during the study are available from the corresponding author on reasonable request.
CONFLICT OF INTEREST
There is no potential conflict of interest related to this work.
AUTHOR CONTRIBUTIONS
Conceptualization: Kee Don Choi. Data curation: Jun-young Seo. Formal analysis: Jun-young Seo and In Hye Song. Investigation: Jun-young Seo. Methodology: Kee Don Choi. Writing - original draft: Jun-young Seo. Writing - review & editing: Hee Kyong Na, Ji Yong Ahn, Jeong Hoon Lee, Kee Wook Jung, Do Hoon Kim, Ho June Song, Gin Hyug Lee, and Hwoon-Yong Jung.
FUNDING STATEMENT
None.
Acknowledgements
None.