 |
 |
- Search
| Korean J Helicobacter Up Gastrointest Res > Volume 21(1); 2021 > Article |
|

Abstract
Background/Aims
Methods
Results
Conclusions
Supplementary Materials
Supplementary Fig. 3.
Fig. 1.
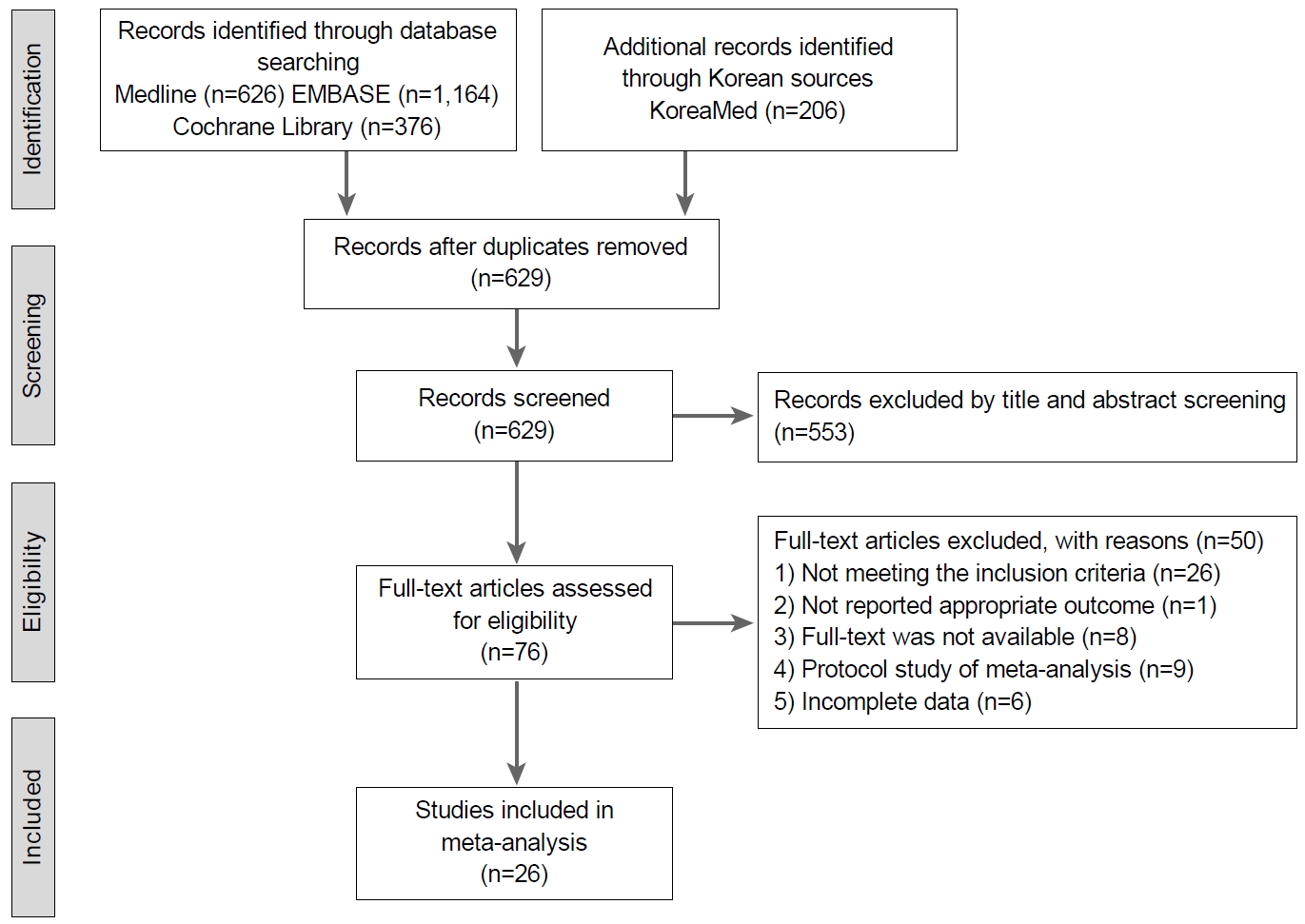
Fig. 2.
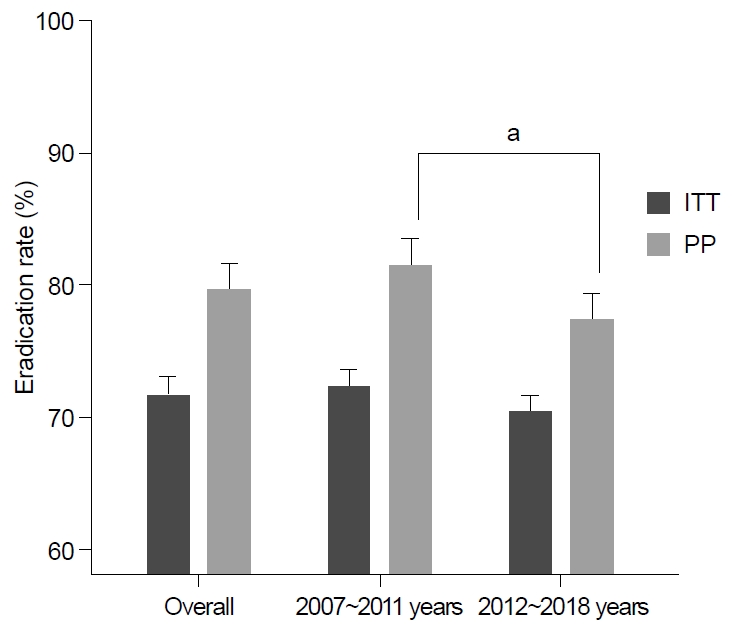
Fig. 3.
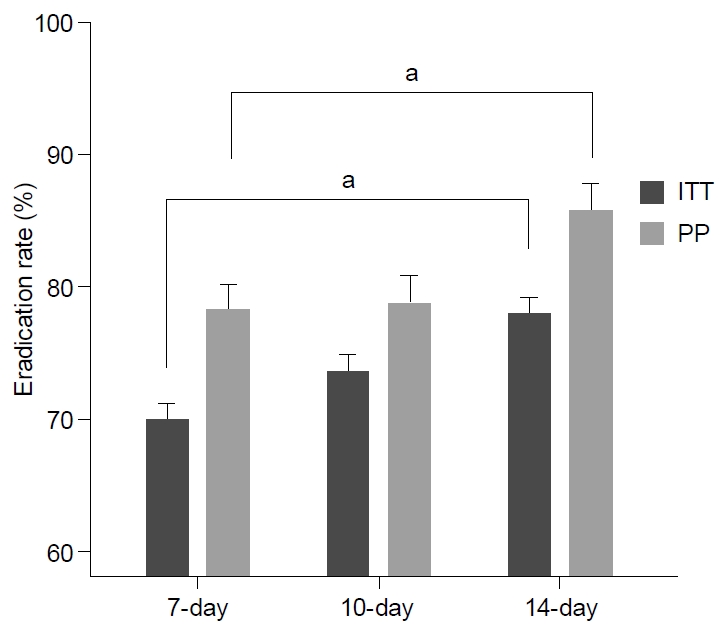
Fig. 4.
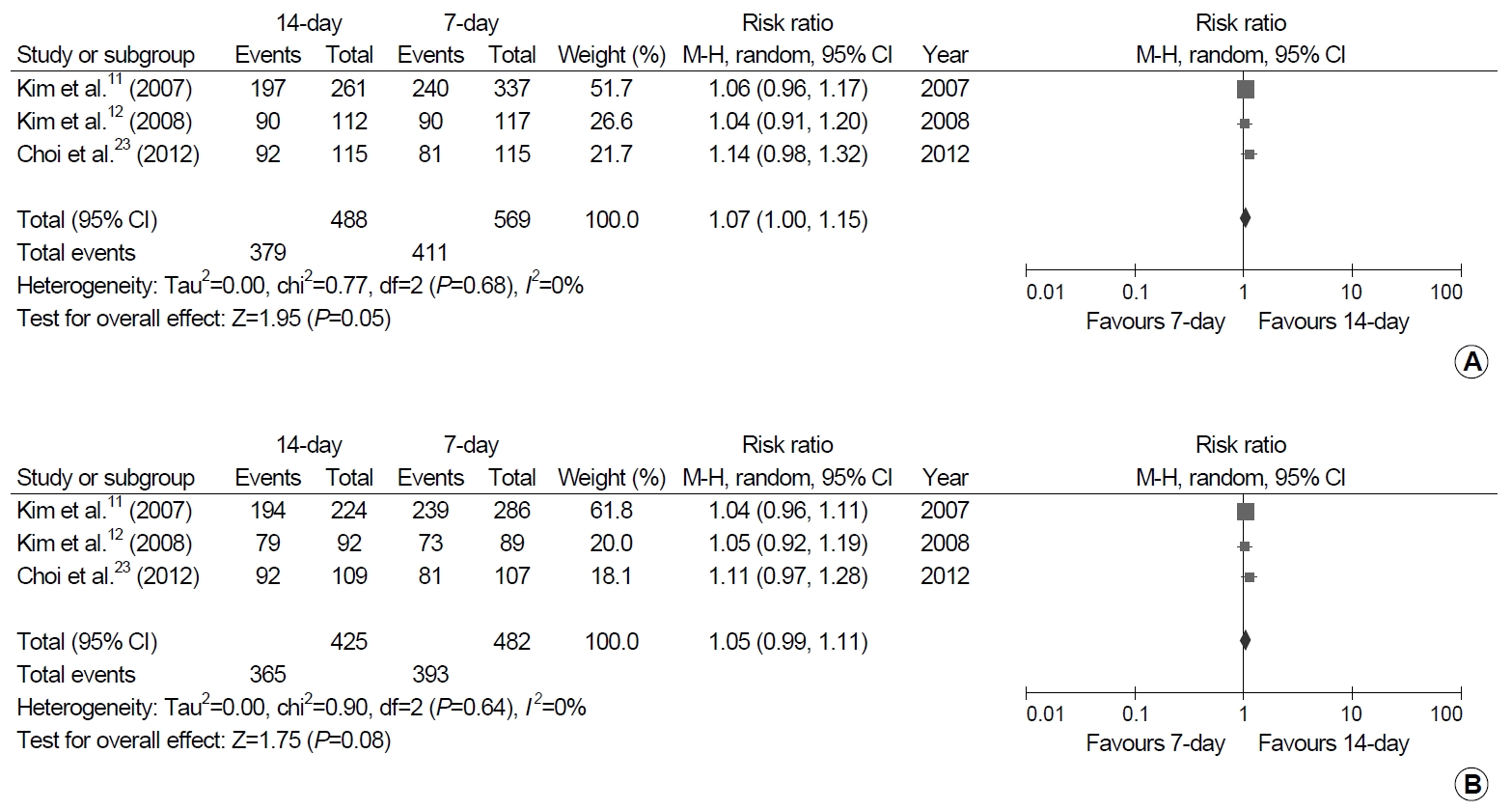
Fig. 5.
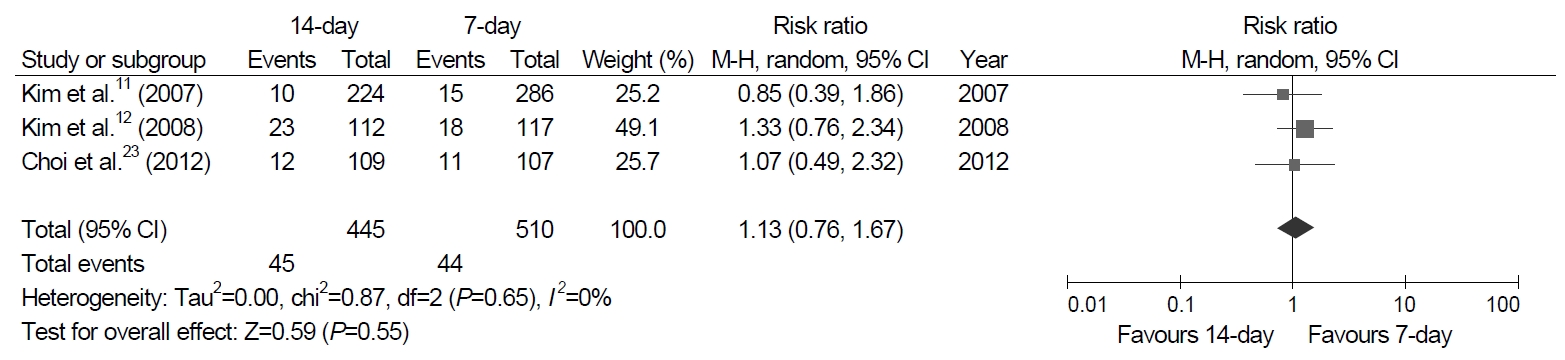
Table 1.
| Study | Study period | Indication of eradication | Treatment arms (number of patients) | Confirmation of eradication | Eradication rate ITT | Eradication rate PP |
|---|---|---|---|---|---|---|
| Choi et al. [10] (2007) | Jan 2006 to Dec 2006 | Dyspepsia and others with H. pylori | TTa 7-day (576) | CLO, histology | 68.4% (394/576) | 82.1% (394/480) |
| Kim et al. [11] (2007) | Dec 2002 to May 2003 | Peptic ulcer including scar with H. pylori | TT 7-day (337) | UBT | TT 7-day: 71.2% (240/337) | TT 7-day: 83.6% (239/286) |
| TT 14-day (261) | TT 14-day: 75.5% (197/261) | TT 14-day: 86.6% (194/224) | ||||
| Choi et al. [14] (2008) | Jan 2007 to Dec 2007 | H. pylori positive patients | TT 7-day (81) | UBT | TT 7-day: 71.6% (58/81) | TT 7-day: 76.6% (58/76) |
| STb 10-day (77) | ST 10-day: 77.9% (60/77) | ST 10-day: 85.7% (60/70) | ||||
| Jung et al. [16] (2008) | May 2004 to Nov 2005 | Active peptic ulcer with H. pylori | TT 7-day (12) | CLO, histology | TT 7-day: 66.7% (8/12) | TT 7-day: 80.0% (8/10) |
| Kim et al. [13] (2008) | Jul 2006 to Feb 2007 | Peptic ulcer with H. pylori | TT 7-day (119) | UBT | TT 7-day: 76.9% (90/117) | TT 7-day: 82.0% (73/89) |
| TT 14-day (112) | TT 14-day: 80.4% (90/112) | TT 14-day: 85.9% (79/92) | ||||
| Kim et al. [12] (2008) | Jun 2005 to Apr 2006 | Dyspepsia and peptic ulcer | TT 14-day (93) | UBT, CLO | TT 14-day: 91.4% (85/93) | TT 14-day: 92.1% (82/89) |
| TT 14day with amcmdUin cjd (93) | TT 14-day with qid: 89.2% (83/93) | TT 14-day with qid: 90.8% (79/87) | ||||
| Kim et al. [15] (2008) | Mar 2006 to Feb 2007 | Dyspepsia and peptic ulcer | TT 7-day (179) | UBT | TT 7-day: 72.1% (129/179) | TT 7-day: 78.7% (129/164) |
| TT 7-day+yogurt (168) | TT 7-day+yogurt: 79.2% (133/168) | TT 7-day+yogurt: 87.5% (133/152) | ||||
| Kim et al. [17] (2008) | May 2006 to Apr 2007 | H. pylori infected patients | TT 7-day (129) | UBT | TT 7-day: 72.1% (93/129) | TT 7-day: 78.8% (93/118) |
| TT 7-day+ecabet (128) | TT 7-day+ecabet: 78.9% (101/128) | TT 7-day+ecabet: 88.6% (101/114) | ||||
| Lee et al. [18] (2010) | May 2006 to Sep 2008 | Peptic ulcer, post-endoscopic resection of adenoma and EGC | TT 7-day (492) | UBT | TT 7-day: 70.7% (348/492) | TT 7-day: 75.2% (348/463) |
| Song et al. [19] (2010) | Jun 2005 to May 2008 | H. pylori infected patients | TT 7-day (331) | UBT | TT 7-day: 71.6% (237/331) | TT 7-day: 80.0% (237/296) |
| TT 7-day+probiotics (330) | TT 7-day+probiotics: 80.0% (264/330) | TT 7-day+probiotics: 85.4% (264/309) | ||||
| TT 7-day+probiotics+mucoprotective (330) | TT 7-day+probiotics+mucoprotective: 82.1% (271/330) | TT 7-day+probiotics+mucoprotective: 84.9% (271/319) | ||||
| Choi et al. [21] (2011) | Jul 2008 to Sep 2009 | H. pylori infected patients | TT 7-day (99) | UBT | TT 7-day: 77.8% (77/99) | TT 7-day: 85.6% (77/90) |
| PPI, amoxicillin, levofloxacin 7-day (98) | PPI, amoxicillin, levofloxacin 7-day: 65.3% (64/98) | PPI, amoxicillin, levofloxacin 7-day: 73.8% (64/87) | ||||
| PPI, amoxicillin, levofloxacin, rifaximin 7-day (98) | PPI, amoxicillin, levofloxacin, rifaximin 7-day: 74.5% (73/98) | PPI, amoxicillin, levofloxacin, rifaximin 7-day: 80.2% (73/91) | ||||
| Kim et al. [20] (2011) | Oct 2008 to Feb 2009 | Dyspepsia and peptic ulcer | TT 14-day (204) | UBT | TT 14-day: 75.0% (153/204) | TT 14-day: 85.0% (153/180) |
| ST 10-day (205) | ST 10-day: 85.9% (176/205) | ST 10-day: 92.6% (176/190) | ||||
| Kim et al. [26] (2012) | May 2009 to Apr 2010 | Peptic ulcer disease | TT 14-day (104) | UBT, CLO | TT 14-day: 74.0% (77/104) | TT 14-day: 82.8% (77/93) |
| DTc 14-day (104) | DT 14-day: 67.3% (70/104) | DT 14-day: 78.4% (69/88) | ||||
| Choi et al. [23] (2012) | Mar 2008 to Aug 2011 | H. pylori-positive gastritis or peptic ulcer disease | TT 7-day (115) | UBT, histology | TT 7-day: 70.4% (81/115) | TT 7-day: 75.7% (81/107) |
| TT 10-day (115) | TT 10-day: 74.7% (86/115) | TT 10-day: 81.9% (86/105) | ||||
| TT 14-day (115) | TT 14-day: 80.0% (92/115) | TT 14-day: 84.4% (92/109) | ||||
| ST 10-day (115) | ST 10-day: 75.6% (87/115) | ST 10-day: 82.0% (87/106) | ||||
| Chung et al. [22] (2012) | Nov 2010 to Aug 2011 | Peptic ulcer disease | TT 10-day (80) | UBT | TT 10-day: 58.7% (47/80) | TT 10-day: 67.6% (46/68) |
| ST 10-day (79) | ST 10-day: 75.9% (60/79) | ST 10-day: 86.8% (59/68) | ||||
| Oh et al. [25] (2012) | Dec 2009 to Dec 2010 | Dyspepsia | TT 7-day (130) | UBT | TT 7-day: 63.0% (82/130) | TT 7-day: 64.5% (82/127) |
| ST 10-day (116) | ST 10-day: 79.3% (92116) | ST 10-day: 81.9% (91/111) | ||||
| Park et al. [24] (2012) | May 2009 to Dec 2010 | H. pylori infected patients | TT 7-day (164) | UBT | TT 7-day: 62.2% (102/164) | TT 7-day: 76.0% (102/125) |
| ST 10-day (162) | ST 10-day: 77.8% (126/162) | ST 10-day: 87.9% (126/132) | ||||
| Kim et al. [28] (2013) | Aug 2009 to Oct 2010 | H. pylori infected pa社ents | TT 7-day (135) | UBT | ST 7-day: 72.6% (98/135) | ST 7-day: 85.2% (98/115) |
| CTd 5-day (135) | CT 5-day: 80.7% (109/135) | CT 5-day: 91.4% (106/116) | ||||
| Lee et al. [27] (2013) | Jul 2011 to Mar 2012 | Peptic ulcer disease | TT 7-day (308) | UBT | TT 7-day: 69.5% (214/308) | TT 7-day: 75.9% (214/282) |
| Tailored therapy (218) | Tailored therapy: 80.7% (176/218) | Tailored therapy: 91.2% (176/193) | ||||
| Heo et al. [29] (2014) | Apr 2012 to Nov 2013 | H. pylori-positive gastritis or peptic ulcer disease | TT 10-day (174) | UBT | TT 10-day: 70.7% (123/174) | TT 10-day: 78.4% (120/153) |
| CT 10-day (174) | CT 10-day: 78.7% (137/174) | CT 10-day: 88.7% (133/150) | ||||
| Lee et al. [31] (2014) | May 2010 to Sep 2013 | Peptic ulcer, dyspepsia, EGC | TT 7-day (115) | UBT, CLO | TT 7-day: 64.3% (74/115) | TT 7-day: 68.5% (74/108) |
| ST 10-day (111) | ST 10-day: 72.1% (80/111) | ST 10-day: 78.4% (80/102) | ||||
| ST 15-day (106) | ST 15-day: 80.2% (85/106) | ST 15-day: 89.5% (85/95) | ||||
| Park et al. [30] (2014) | Nov 2011 to Oct 2012 | EGC, dysplasia | TT 7-day (57) | UBT | TT 7-day: 71.9% (41/57) | TT 7-day: 73.2% (41/56) |
| Susceptibility-guided therapy (57) | Susceptibility-suided therapy: 94.7% (54/57) | Susceptibility-guided therapy: 96.4% (54/56) | ||||
| Bang et al. [33] (2015) | Jun 2012 to Aug 2013 | Peptic ulcer and dyspepsia | TT 7-day (55) | UBT | TT 7-day: 76.4% (42/55) | TT 7-day: 87.5% (42/48) |
| TT 7-day+pronase (57) | TT 7-day+pronase: 56.1% (32/57) | TT 7-day+pronase: 68.1% (32/47) | ||||
| Lee et al. [32] (2015) | Jul 2013 to Mar 2014 | H. pylori-positive gastritis or peptic ulcer disease | TT 7-day | UBT | TT 7-day: 64.1% (109/170) | TT 7-day: 76.2% (109/143) |
| PAM 7-day | PAM 7-day: 68.8% (117/170) | PAM 7-day: 84.2% (117/139) | ||||
| ST 10-day | ST 10-day: 70.7% (119/170) | ST 10-day: 84.4% (119/141) | ||||
| CT 7-day | CT 7-day: 79.4% (135/170) | CT 7-day: 94.4% (135/143) | ||||
| Chung et al. [34] (2016) | May 2013 to Mar 2015 | Peptic ulcer, EGC, MALT lymphoma | TT 10-day (171) | UBT | TT 10-day: 83.0% (142/171) | TT 10-day: 82.8% (106/128) |
| ST 10-day (170) | ST 10-day: 88.8% (151/170) | ST 10-day: 89.5% (119/133) | ||||
| CT 10-day (176) | CT 10-day: 93.2% (164/176) | CT 10-day: 94.4% (135/143) | ||||
| Kim et al. [35] (2016) | Mar 2011 to Sep 2014 | Peptic ulcer, EGC, MALT lymphoma | TT 7-day (295) | UBT, CLO, histology | TT 7-day: 70.8% (209/295) | TT 7-day: 76.9% (206/268) |
| ST 10-day (306) | ST 10-day: 82.4% (252/306) | ST 10-day: 88.8% (247/278) |
ITT, intention-to-trat; PP, per protocol; H. pylori, Helicobacter pylori; TT, standard triple therapy; CLO, Campylobacter like organism test; UBT, urea breath test; ST, sequential therapy; qid, four times per day; EGC, early gastric cancer; PPI, proton pump inhibitor; DT, dual therapy; CT, concomitant therapy; PAM, proton pump inhibitor+amoxicillin+metronidazole; MALT, mucosa-associated lymphoid tissue.
Table 2.
| Study | Regimens | Overall adverse event | Severe adverse event | Most common adverse event |
|---|---|---|---|---|
| Choi et al. [10] (2007) | TTa 7-day | 18.1% (87/480) | N/A | Bitterness (7.1%, 34/480) |
| Kim et al. [11] (2007) | TT 7-day | 5.1% (15/286) | N/A | Diarrhea, loose stool (3.0%, 9/286) |
| TT 14-day | 4.6% (10/224) | Diarrhea, loose stool (2.3%, 5/224) | ||
| Choi et al. [14] (2008) | TT 7-day | 29.6% (24/81) | N/A | Loose stool (11.1%, 9/81) |
| STb 10-day | 28.6% (22/77) | Taste alteration (7.8%, 6/77) | ||
| Jung et al. [16] (2008) | TT 7-day | 8.3% (1/12) | N/A | Taste disturbance (8.3%, 1/12) |
| Kim et al. [13] (2008) | TT 7-day | 15.4% (18/117) | Nausea/vomiting, diarrhea (6.7%, 8/117) | |
| TT 14-day | 20.5% (23/112) | Diarrhea (6.6%, 11/112) | ||
| Kim et al. [12] (2008) | TT 14-day | 30.1% (28/93) | N/A | Bitter taste 21.5% (20/93) |
| TT 14-day with amoxicillin qid | 39.8% (37/93) | Bitter taste 22.6% (21/93) | ||
| Kim et al. [15] (2008) | TT 7-day | 26.3% (47/179) | 3.3% (6/179) | Diarrhea 7.8% (14/179) |
| TT 7-day+yogurt | 41.1% (69/168) | 5.3% (9/168) | Metallic taste 16.7% (28/168) | |
| Kim et al. [17] (2008) | TT 7-day | 21.8% (26/129) | N/A | Metallic taste 10.9% (14/129) |
| TT 7-day+ecabet | 20.3% (24/128) | Dizziness 4.7% (6/128) | ||
| Lee et al. [18] (2010) | TT 7-day | N/A | N/A | N/A |
| Song et al. [19] (2010) | TT 7-day | 19.0% (63/331) | N/A | Diarrhea 6.0% (20/331) |
| TT 7-day+probiotics | 14.5% (48/330) | Taste disturbance 4.2% (14/330) | ||
| TT 7-day+probiotics+mucoprotective | 9.1% (30/330) | Diarrhea 3.0% (10/330) | ||
| Choi et al. [21] (2011) | TT 7-day | 32.2% (29/90) | N/A | Diarrhea 16.7% (15/90) |
| PPI, amoxicillin, levofloxacin 7-day | 19.5% (17/87) | Diarrhea 11.5% (10/87) | ||
| PPI, amoxicillin, levofloxacin, rifaximin 7-day | 9.9% (37/91) | Diarrhea 3.3% (3/91) | ||
| Kim et al. [20] (2011) | TT 14-day | 13.3% (24/180) | N/A | Diarrhea 6.1% (11/180) |
| ST 10-day | 18.9% (36/190) | Diarrhea 6.8% (13/190) | ||
| Kim et al. [26] (2012) | TT 14-day | 35.6% (37/104) | N/A | Bitter taste 26.9% (28/104) |
| DTc 14-day | 18.3% (19/104) | Bitter taste 4.8% (5/104) | ||
| Choi et al. [23] (2012) | TT 7-day | 10.3% (11/107) | N/A | Loose stool, epigastric discomfort 2.8% (3/107) |
| TT 10-day | 13.3% (14/105) | Nausea/vomiting 3.8% (4/105) | ||
| TT 14-day | 11.0% (12/109) | Nausea/vomiting, epigastric discomfort 2.8% (3/109) | ||
| ST 10-day | 14.2% (15/106) | Nausea/vomiting 3.8% (4/106) | ||
| Chung et al. [22] (2012) | TT 10-day | 26.3% (21/80) | N/A | Diarrhea 17.5% (14/80) |
| ST 10-day | 29.1% (23/79) | Diarrhea 21.5% (17/79) | ||
| Oh et al. [25] (2012) | TT 7-day | 23.8% (31/130) | N/A | Epigastric soreness 5.4% (7/130) |
| ST 10-day | 27.5% (32/116) | Diarrhea, epigastric soreness 6.0% (7/116) | ||
| Park et al. [24] (2012) | TT 7-day | 25.5% (35/137) | N/A | Diarrhea 7.3% (10/137) |
| ST 10-day | 28.0% (40/143) | Diarrhea 4.9% | ||
| Kim et al. [28] (2013) | ST 7-day | 25.2% (34/135) | N/A | Bitter taste 20.0% (27/135) |
| CTd 5-day | 35.6% (48/135) | Bitter taste 27.4% (37/135) | ||
| Lee et al. [27] (2013) | TT 7-day | 26.8% (112/418) | N/A | Bitter taste 12.9% (54/418) |
| PAM 7-day | 29.3% (98/334) | Nausea 5.7% (19/334) | ||
| Heo et al. [29] (2014) | TT 10-day | 34.0% (55/162) | N/A | Taste disturbance 17.3% (28/162) |
| CT 10-day | 38.3% (62/162) | Diarrhea 25.3% (41/162) | ||
| Lee et al. [31] (2014) | TT 7-day | 29.6% (34/115) | 9.6% (11/115) | Heartburn 9.6% (11/115) |
| ST 10-day | 31.5% (35/111) | 12.6% (14/111) | Heartburn 12.6% (14/111) | |
| ST 15-day | 32.1% (34/106) | 13.2% (14/106) | Heartburn 11.3% (12/106) | |
| Park et al. [30] (2014) | TT 7-day | 8.8% (5/57) | N/A | Diarrhea 3.5% (2/57) |
| Susceptibility-guided therapy | 7.0% (4/57) | Nausea/vomiting 3.5% (2/57) | ||
| Bang et al. [33] (2015) | TT 7-day | 39.6% (19/48) | N/A | Bitter taste 29.2% (14/48) |
| TT 7-day+pronase | 48.9% (23/47) | Bitter taste 40.4% (19/47) | ||
| Lee et al. [32] (2015) | TT 7-day | 50.6% (86/170) | N/A | Bitter taste 16.5% (28/170) |
| PAM 7-day | 48.2% (82/170) | Nausea 18.8% (32/170) | ||
| ST 10-day | 41.2% (70/170) | Nausea 11.8% (20/170) | ||
| CT 7-day | 40.6% (69/170) | Epigastric pain 12.4% (21/170) | ||
| Chung et al. [34] (2016) | TT 10-day | 35.1% (60/171) | N/A | Taste disturbance 21.6% (37/171) |
| ST 10-day | 32.4% (55/170) | Taste disturbance 19.4% (33/170) | ||
| CT 10-day | 39.2% (69/176) | Taste disturbance 25.0% (44/176) | ||
| Kim et al. [35] (2016) | TT 7-day | 43.0% (122/284) | N/A | Bitter taste 28.5% (81/284) |
| ST 10-day | 44.4% (132/297) | Bitter taste 24.2% (72/297) |
TT, standard triple therapy; N/A, not applicable; ST, sequential therapy; qid, four times per day; PPI, proton pump inhibitor; DT, dual therapy; CT, concomitant therapy; PAM, proton pump inhibitor+amoxicillin+metronidazole.
REFERENCES
- TOOLS
-
METRICS

-
- 1 Crossref
- 4,436 View
- 146 Download
- Related articles in Korean J Helicobacter Up Gastrointest Res
-
The Eradication Rate of Helicobacter pylori Using PPI-based Triple Therapy in Jeju Island2009 ;9(1)
Critical Appraisal of Systematic Review/Meta-analysis2015 June;15(2)






